Reliable PCBs
– because failure is not an option.
We produce PCBs for demanding customers
Our ongoing goal is to assist our customers achieve the best possible time to market and competitive advantage by producing printed circuit boards in a sustainable way at the lowest total cost through our competence, delivery accuracy and product quality.
Your key benefits of working with us



Our latest news

News
NCAB Group Interim report Q1 2024

Blog
PCB Solder mask

Newsletters
Ultra HDI PCBs put high demands on technology and manufacturing
Gold Medal for our sustainability work
NCAB has been awarded with Gold EcoVadis Medal. This result places us amongst the top 5 percent of the 85,000 companies globally that have been assessed by EcoVadis.


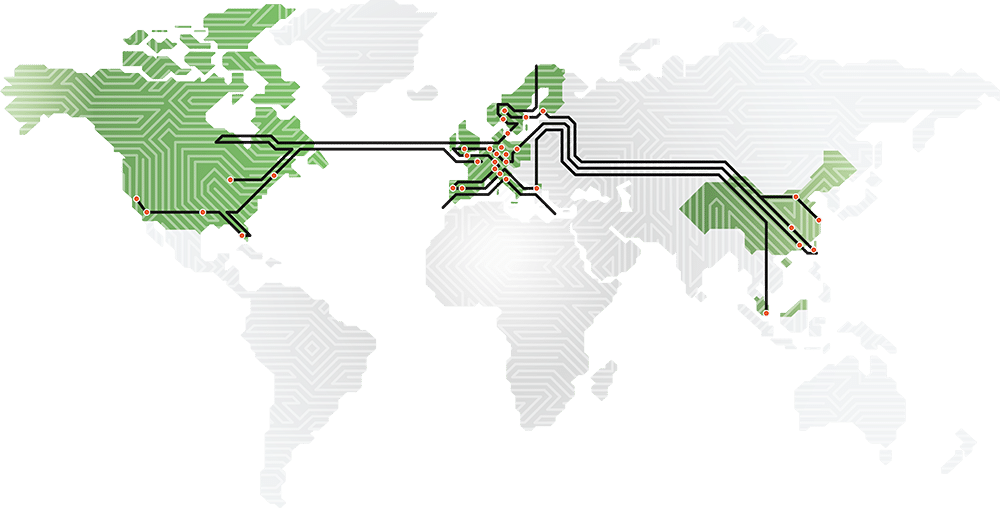
Printed circuit boards from design to volume production
A circuit board is the heart of all electronic equipment. Often it is a complex product. We seek to be at the forefront of PCB technology development and we actively seek cooperation beginning in the design phase. We believe that the best PCB solutions can only be developed through close interaction with our customers.
Our PCB Portfolio ranges from Multilayer, Flexible, Rigid- and Semi-Flex to IMS, RF and HDI printed circuit boards.
17
Companies
32
Factories
45
Markets
603
Specialists
325
Million PCBs produced per year
Last update February 2024

Sustainable business
– our responsibility
Industries we serve
Aerospace, Automotive, Datacom, Defense, Industrial, Medical, Power/Energy, Railway, Safety critical and Telecom









We serve OEM, ODM & EMS customers
Our customers are the reason we exist. NCAB Group is proud to present some of them here.


























Join our team of specialists
Curious about NCAB and reliable PCBs?
Contact us if you have any questions regarding our products or services. We look forward to hearing from you.